Mercury is metallic element that exists in liquid form. Most people will be familiar with it as the substance used in thermometers, barometers, or children’s maze games. Mercury has fascinated humans for much of history. Its elemental symbol, Hg, comes from the Latin hydrargyrum which means liquid silver. As it forms amalgams with other metals, mercury played a central role in the experiments of alchemists as they tried to use it to convert other metals it into gold. Since then, the element has been used for a variety of purposes: in mining, hatting, dentistry, medicine, and many industrial processes. Mercury, particularly as part of organic compounds, is also a potent neurotoxin. In the eighteenth and nineteenth centuries, hatters used a mercury compound to separate animal fur from the pelt. In the process, they inhaled the highly toxic fumes, leading to convulsions, hallucinations, and dementia. Thus originated the phrase “mad as a hatter” (1).
Although mercury is a naturally occurring element (it makes up 0.000008 percent of the earth’s crust) and has natural sources of emission, most notably volcano eruptions, the increased mercury levels of the past 150 years are almost entirely human-caused. Ice cores from the Wind River mountain range in Wyoming produced the following pattern in atmospheric mercury concentrations (2):
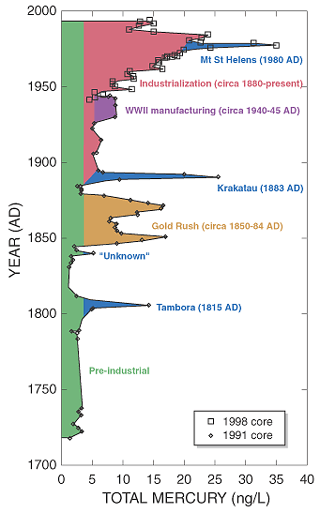
The base concentration from natural emissions is shown in green, with spikes emanating from large volcanic eruptions highlighted in blue. The large increase in concentration of the mid nineteenth century was a result of the Gold Rush period. As mentioned earlier, mercury easily forms amalgams with other metals, effectively dissolving them. Miners used mercury to extract gold from rock, later separating the mercury and gold through filtering and distillation. The red area of the chart shows the effects of industrialization. The most significant source of atmospheric mercury in the industrial period was the burning of fossil fuels, particularly coal. As human demand for cheap energy has risen, so too has the concentration of mercury in the environment.
In the United States , coal-fired power plants are the leading source of mercury emissions. In 2005, they accounted for over 40 percent of all mercury emitted, producing a total of 48 tons (3). To reduce environmental mercury concentrations, the Bush administration instituted a cap-and-trade system, based upon the successful system for reducing the sulfur dioxide emissions that led to acid rain. The plan called for a reduction in power plan mercury emission to 38 tons per year by 2010, and a further reduction to 33 tons by 2018 (3). Under the cap-and-trade system, power plants that reduce their mercury emissions below target levels can sell their pollution credit to a power plant that does not meet pollution targets. Economic theory shows that such a system reduces total emissions in a more cost effective manner than simply capping emissions from each individual power plant.
Environmentalists and scientists have several major complaints about the proposed plan. In general some groups are not ecstatic about companies trading in a major neurotoxin. The biggest concern, however, comes from the major differences between the impacts of sulfur oxides and mercury. Sulfur oxides are relatively light, spread easily, and do not remain long in the environment. As a result, sulfur from a single plant can affect a wide area for a brief period. Mercury, in contrast, is heavier and does not spread as widely. It also remains in the environment for a longer period of time. Therefore, coal plants that choose to buy mercury credit rather than reduce emissions can create “hot spots” of high mercury concentration (4). These “hot spots” greatly increase risk of toxic exposure to local human and wildlife populations.
One of the most widely publicized consequences of elevated mercury concentrations is its affect on seafood. Microscopic aquatic organisms convert anthropogenically-emitted elemental mercury to its highly toxic methylated form. The toxic mercury is transferred to fish and shellfish that feed on these organisms and then continues up the food chain. As most species of fish are slow to expunge acquired mercury, methyl mercury concentrations increase at each level of the food chain in a process known as bioaccumulation. Given the severe consequences of significant exposure to methyl mercury, particularly to developing fetuses, the FDA recommends that pregnant women abstain from eating large predatory fish and that they limit themselves to two servings of fish or shellfish per week (5).
One of the most deadly environmental disasters of the twentieth century occurred as a result of methyl mercury in seafood. In 1956, residents of Minamata , Japan began being hospitalized with symptoms of an unknown neurological disease. They complained of tremors, numbness, muscle weakness, reduced vision, and damage to hearing and speech. A lengthy investigation revealed that they were suffering from severe mercury poisoning as a result of eating contaminated seafood. The source of the mercury was the Chisso Corporation’s chemical factory, which hand dumped contaminated wastewater into Minamata Bay since 1932 (6).
In 1996, the Japanese government offered sufferers of Minamata disease compensation. As of 2001, it had certified 2,264 victims of whom 1,453 had already died. At that time, 17,128 more had applied for recognition with perhaps an additional 20,000 eligible (7).
References